DOI:10.1021/acs.biomac.2c01474
- 1. Institute for Advanced Biosciences, Keio University
- 2. Graduate School of Media and Governance, Keio University
- 3. Spiber Inc.
- 4. Japan Synchrotron Radiation Research Institute
- 5. Biomacromolecules Research Team, RIKEN Center for Sustainable Resource Science
- 6. Department of Material Chemistry, Kyoto University
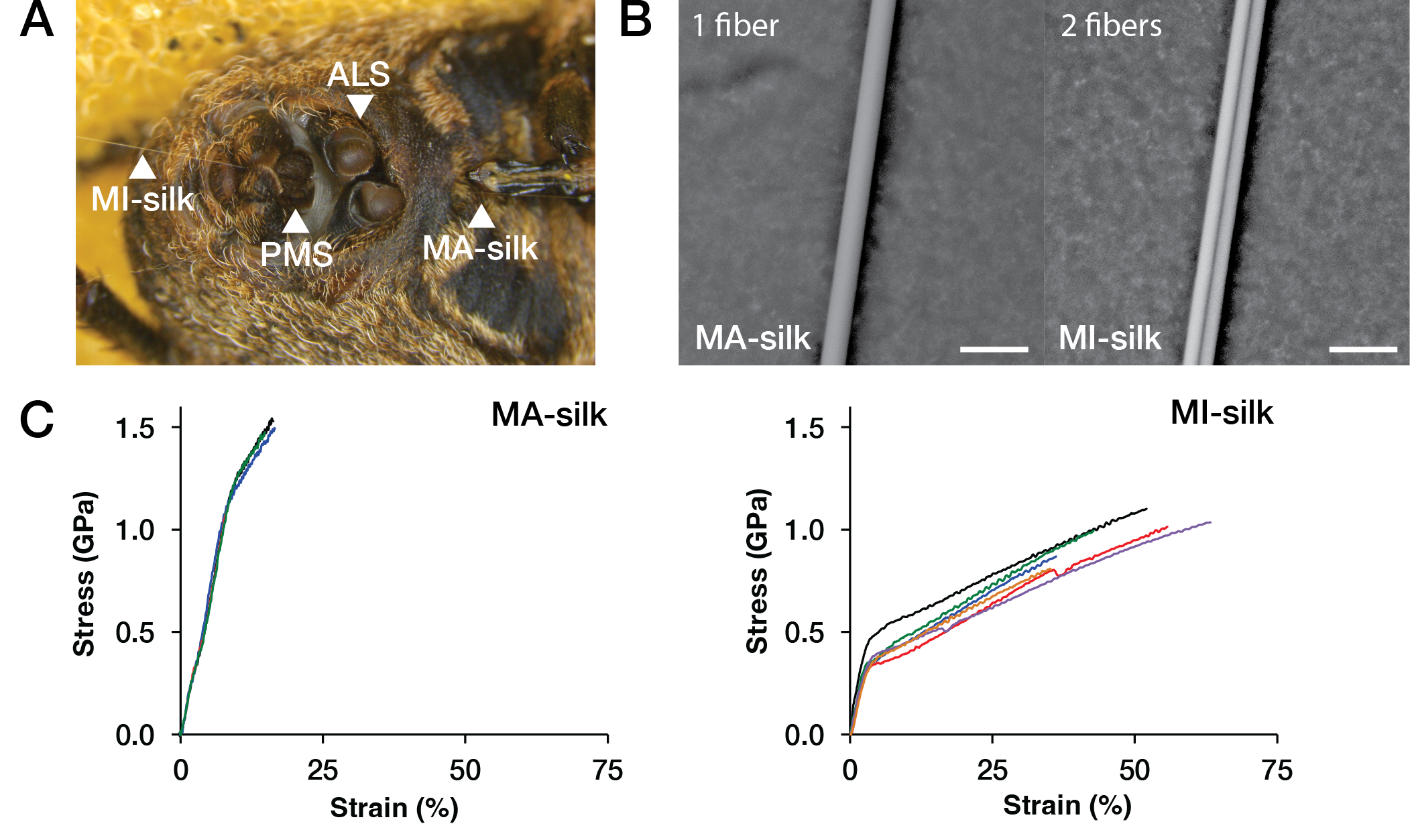
From “Composition of Minor Ampullate Silk Makes Its Properties Different from Those of Major Ampullate Silk”
Abstract
Spider’s minor ampullate silk, or MI-silk, exhibits distinct mechanical properties and water resistance compared to its major ampullate counterpart (MA-silk). The principal protein constituent of MI-silk is known as minor ampullate spidroin, or MiSp, and while its sequence has been deciphered and is thought to underlie the differences in properties with MA-silk, the composition of MI-silk and the relationship between its composition and properties remain elusive. In this study, we set out to investigate the mechanical properties, water resistance, and proteome of MA-silk and MI-silk from Araneus ventricosus and Trichonephila clavata. We also synthesized artificial fibers from major ampullate spidroin, MaSp1 and 2, and MiSp to compare their properties. Our proteomic analysis reveals that the MI-silk of both araneids is composed of MiSp, MaSp1, and spidroin constituting elements (SpiCEs). The absence of MaSp2 in the MI-silk proteome and the comparison of the water resistance of artificial fibers suggest that the presence of MaSp2 is the reason for the disparity in water resistance between MI-silk and MA-silk.